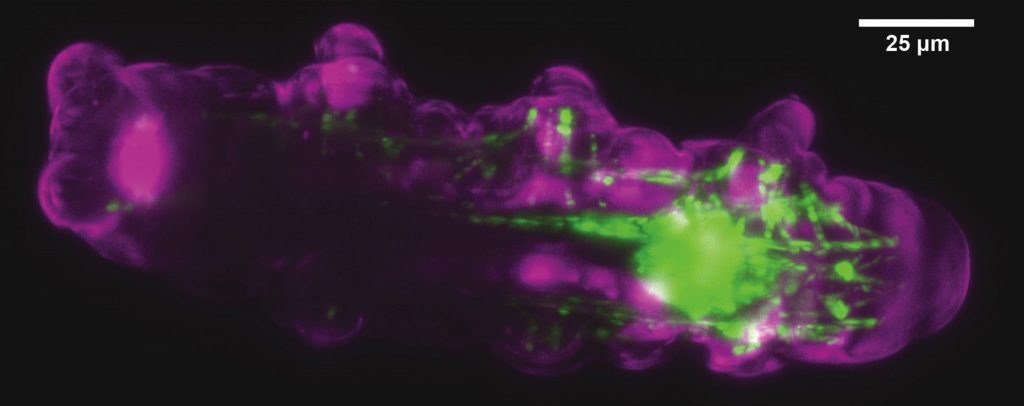
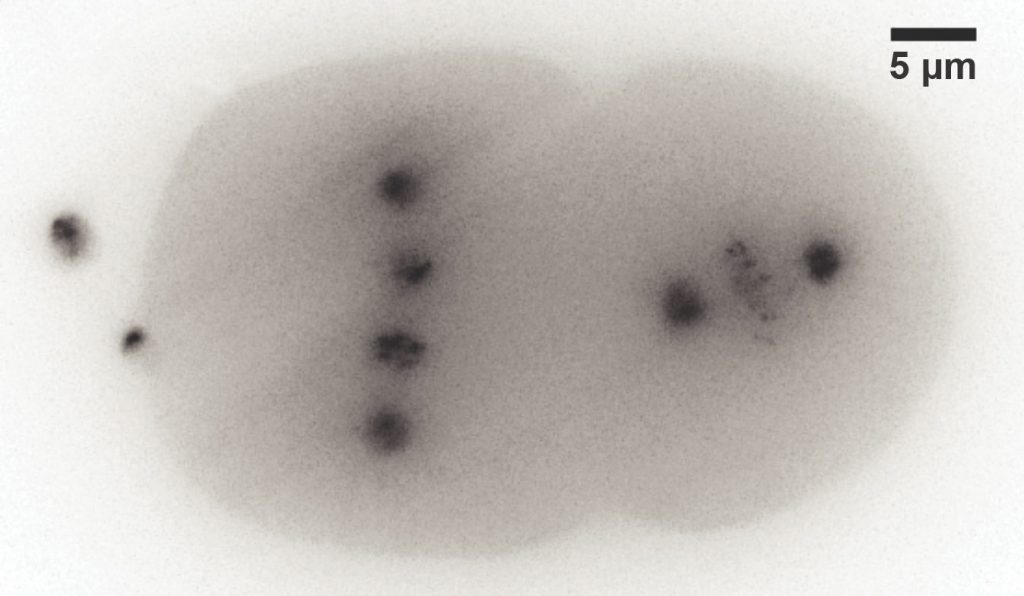
Mizar TILT
Light Sheet Conversion System for Fluorescence Microscopes from Mizar Imaging
The Mizar TILT is a light sheet illumination system for fluorescence microscopes. It provides all the advantages of light sheet imaging with minimal photodamage, bleaching, or toxicity. It allows imaging of epifluorescence with high‐magnification and high‐numerical aperture objectives for maximum resolution, and supports all imaging modalities including widefield fluorescence and DIC.

The TILT uses a cylindrical lens to create a light sheet from an expanded laser source, in a manner similar in part to that of other light sheet systems. The critical difference is the TILT directs the light sheet at an angle — hence the tilt in TILT — to focus the thinnest part of the light sheet as close as possible to the objective lens. This allows researchers to use high‐NA objectives, and to reap the consequent benefits of efficient photon capture and maximal resolution.

| Feature | Benefit |
| Imaging modalities | Extended‐duration imaging |
| High‐frequency capture | |
| Imaging of photosensitive process | |
| Imaging in multiple colors | |
| Light sheet illumination | Minimal‐to‐no photodamage or bleaching |
| Optimal function even with high‐NA objectives | Efficient photon capture |
| Maximal resolution | |
| Support for multiple manufacturers | Installs on the microscope of your choice |
| Works without post‐imaging computational processing | Native imaging |
| Ease of use |
The Mizar TILT is a product of Mizar Imaging, LLC. It is engineered by Cairn Research Ltd..
Frequently Asked Questions
What is the Mizar TILT?
The Mizar TILT is a light sheet conversion system for fluorescence microscopes. It provides bona fide light sheet illumination capabilities and inherent advantages (e.g., limited photodamage), along with the advantages of epifluorescence such as high‐resolution imaging.
Does the TILT work in the same way as other light sheets?
In part. The Mizar TILT uses a cylindrical lens to create a light sheet from an expanded laser source, similar to the way some do. The critical difference is the TILT directs the light sheet at an angle — hence the tilt in TILT — to focus the thinnest part of the light sheet as close as possible to the objective lens. This allows the researcher to use high‐NA objectives, and to reap the consequent benefits of efficient photon capture and maximal resolution.
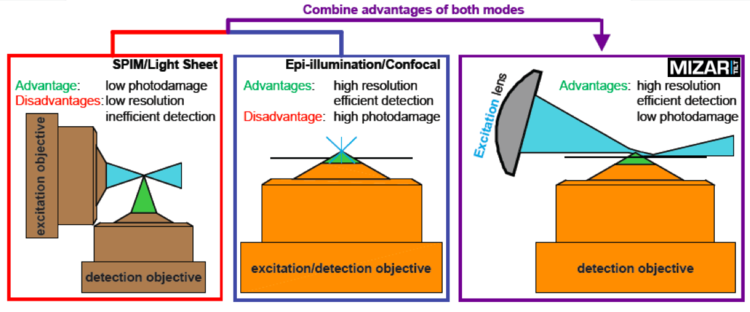
So the TILT allows us to use high‐NA objectives. Why is that so important?
That allows more efficient light‐gathering and higher resolution. Though high‐numerical‐aperture objectives might not be appropriate for all applications, they are essential for others because of these two related advantages. While gaining higher resolution is a reward all its own, consider also how an increased efficiency in photon capturing ability opens avenues to new experiments: not only does increased efficiency imply greater sensitivity for observing low‐abundance molecules, it also implies fewer, potentially damaging, excitation photons will be required to pass a threshold of detection. It follows that the TILT may have even fewer photodamaging and photobleaching effects than other light sheets that are restricted to lower‐NA objectives, though Mizar Imaging have not proven this.
How else does the TILT differ?
One of the key benefits to the TILT is its simplicity. Indeed, it is essentially no more difficult to use than a standard epifluorescence rig. Because the TILT attaches to virtually any microscope stand, you can use it on a scope you already work with on a daily basis. No dedicated operator is needed. No post‐imaging computational processing is required. Just mount your sample, adjust the beam, and you are ready to create killer images.
What does the TILT include?
The TILT itself is composed of the TILT illuminator and a substage with encoded X–Y axis control and piezoelectronic Z‐axis control.
What equipment would the user need?
The TILT in essence acts as the stage on your microscope rig — so you would need everything else, including the laser, which is the light source for the TILT.
Does the TILT include a laser?
Researchers will have specific requirements for which lasers they need, depending on which fluorophores they are using. It would therefore not make sense to try to include all the various wavelength lasers researchers could possibly need. BioVision Technologies can supply lasers and laser combiners as needed.
Sounds pretty clever. Who designed the TILT?
A researcher: Paul Maddox, the founder of Mizar Imaging, in his lab at The University of North Carolina.
What kind of laser is required for the TILT to work?
Mizar Imaging finds that a 100 mW laser at 50%–75% power meets most if not all of their needs and would feel comfortable using a 50 mW laser as well.
How does the TILT achieve high resolution images, while other light sheets don’t?
The key to the TILT is in the tilt. The TILT light sheet comes in at an angle so that the narrowest part of the sheet is substantially closer to the objective lens. Because of this, the user can image with high‐numerical‐aperture objectives. And because of that, the researcher can image at much higher resolution and with far greater levels of detection sensitivity.
Is the TILT easy to use?
Yes. Like any precision research instrument — such as the microscope itself — the TILT does require some training and knowledge of the underlying principles to function optimally, but it is easy to get up to speed; with that accomplished use is a straightforward matter of adjusting the sheet during image acquisition.
Do you provide training?
Yes, BioVision Technologies provides training and follow up support.
Does imaging with the TILT require post‐imaging computational processing?
No. You can capture beautiful native images and image sequences (i.e. movies) and will not need special computational processing to determine what you have. You can know right when you grab them how good and publication‐worthy they are.
So the TILT light sheet works by coming in at an angle. What angle is that?
Mizar Imaging finds that generally an angle between 2.5° and 4.5° works best. The angle is simple to adjust, and different angles may work better for different kinds of biological samples. With a little bit of practice, one can become adept at focusing the sheet and experimenting at various angles to meet your needs. In general, too shallow an angle can lead to “shadowing” (see below), while too steep an angle could lead to scattered light entering the objective lens (see “light scatter” below).
Can one adjust the angle of the TILT light sheet?
Yes, one can and one should. BioVision Technologies will train you how to focus the light sheet and adjust its angle. Typically, Mizar Imaging finds that they do not need to adjust the angle of the sheet much, but it is possible some kinds of samples may work best at various angles.
Do steep angles of the TILT light sheet cause artifacts?
High angles can cause a wide‐angle light scattering effect as excitation light enters the objective, but this can be removed with proper emission filters.
Does the TILT cause shadowing artifacts as seen with other light sheets?
Shadowing can be a problem at lower angles, but adjusting the TILT so the light sheet comes in a slightly higher angles can ameliorate and even remove this effect. As with all imaging, a lot depends on the sample imaging and on preparation of the sample.
Does it cause photodamage / photobleaching / phototoxicity?
Not that Mizar Imaging has seen so far, but the company acknowledges that the TILT is in its early days. The company will keep researchers posted as it tests the limits. The company is just as interested as are researchers in seeing how much it pushes experiments into new areas of duration and high‐frequency sampling to gain better temporal resolution. The company will also be testing the boundaries of photobleaching using low abundance factors. The company will pin posts to its Twitter feed as it reaches new milestones, and encourages researchers to check there for updates.
What are the extremes of photodamage and bleaching that Mizar Imaging has tested?
As of this writing, the company has imaged cells in culture under continuous illumination for 24 hours without seeing deleterious effects, however, the samples under test are varied and the boundaries being tested are moving too fast to keep updated here. The company will update its Twitter feed as it and other users explore the new light sheet freedom affored by the TILT. Please check there to follow developments.
Can I use the TILT with my existing microscopes?
Yes. If you use a microscope stand from any of the major microscope companies (Zeiss, Leica Microsystems, Nikon Instruments, Olympus), then the TILT will fit onto it without further modifications. If for some unforseen reason it does not fit, Mizar Imaging will modify the TILT stage to make it fit.
Can I do live cell imaging?
Yes, by all means. The TILT is optimized for live cell imaging.
Can I do high‐frequency imaging?
Yes! Research goals often call for sampling at high temporal resolution. You can use the TILT for high‐frequency sampling and even continuous illumination/sampling without photodamage.
Which fluorophores does it work with?
All of them. If you have the excitation light source, and the appropriate emission filters, the TILT should be able to create a light sheet for your purposes.
Can I do multi‐channel imaging?
Yes. For your specific applications, contact BioVision Technologies for suggestions and advice based on our experience.
What field of view can the TILT capture?
The TILT light sheet itself does not limit the field of view available. This will depend on the objective lens you choose to use.
Applications
- Developmental Biology
- C. elegans
- D. melanogaster
- D. rerio
- X. laevis
- Embryogenesis
- Intercellular Dynamics
- High‐resolution Imaging of Cleared Tissues
- Imaging of Photosensistive Cells and Tissues
- Plant Imaging
"LITE microscopy: a technique for high numerical aperture, low photobleaching fluorescence imaging" by Tanner C. Fadero et al.. Paper describing Lateral Interference Tilted Excitation (LITE), a novel method of light sheet fluorescence microscopy, which the TILT implements.
Mizar Imaging Twitter feed Mizar Imaging's postings on new milestones reached as it tests the limits of the TILT.